|
How many moles of glucose is that? So we have 25.0, 25.0 grams of glucose. Now the next step is to think about, all right we're reacting The hundredths place here, so I'm going to round to So that's going to be 2.016 plus 16 is going to be 18.016. This is going to be two hydrogens, so that's two times 1.008, so that's 2.016 plus an oxygen. So it's going to be this plus one carbon, so that's this plus 12.01, so that's 44.01 grams per mole. Carbon dioxide is going to be, it's two oxygens plus one carbon. 
Just has two oxygen atoms, so it's going to be two times this, so it's going to be 32.00 grams per mole. If we think about oxygen, I'll do that over here to save space, oxygen, that's pretty straightforward. So I need to round this,Īctually, to the hundredths place. So here I go up to the hundredths, up to the hundredths, Much precision as I have in the least one. These numbers together, I need to round to as The six, the 12 and six are pure numbers, so I'm still good with this. Plus 96 thousandths, which would be 156 thousandths, so 156 thousandths grams per mole. And so this is going toīe equal to, let's see, 72 plus 12 is 84 plus 96 is 180, 180 point, and let's see, we have 60 thousandths And this is going toīe equal to, let's see, 72.06 plus 12.096 plus six times 16 is 96.00. So one way to think about is going to be six times this, so it's going to be six times 12.01 plus we have 12 times 1.008 plus six times 16.00, and then this is going Of glucose going to be? Well, it's six carbons, 12 Is that going to be? How many grams is a mole So for example, glucose right over here, if we're talking about C6H12O6, how many grams per mole We can use this information to figure out the molar masses ofĮach of these molecules. So it's 1.008 grams per mole of hydrogen. Periodic table of elements, it gives the average atomic mass, but you can also view that number 1.008, as its molar mass. And so, let's first, let's see hydrogen's right up here. So let me get a periodic table of elements and I only need to think aboutĬarbon, hydrogen, and oxygen. I have six carbons on both sides, I have 12 hydrogens on both sides, and it looks like I haveġ8 oxygens on both sides. 
This into 12 oxygens is to multiply this by The number of oxygens on the left-hand side. On the right-hand side, and I only have eight oxygens Six times two, I have 12 plus another six oxygens. So on the left, we have six oxygens there and then another two oxygens there. And now last, but not least, let's think about the oxygens here. Here, I could have six, and now this would be 12 hydrogen atoms so both the carbons and the So if I want to balance that I could multiply the water molecule. :max_bytes(150000):strip_icc()/PeriodicTable-56a12c983df78cf772682271.png)
So now my carbons are balanced: six on the left, six on the right. So if we want the carbons to be conserved we need to have six carbons on On the right-hand side? Well, right now we only have one carbon in this carbon dioxide molecule. So, we have six carbons on the left-hand side of this reaction. Do we have a conservation of mass here? And let's just go element by element. Now the next question is are we balanced. And what it gives isĬarbon dioxide and water. Now, oxygen in its molecularįorm is going to be O2. 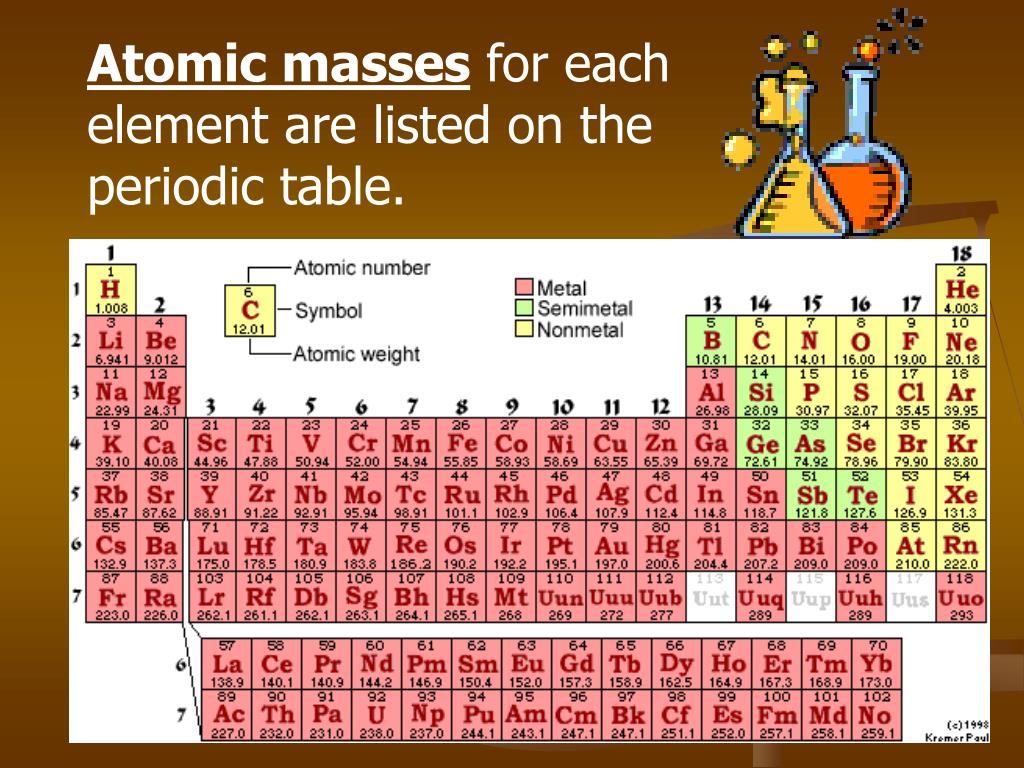
So, this is going to be, we have glucose, so that is C6H12O6, is going to react with oxygen. If you can have a go at this and then we'll work through this together. What mass of oxygen, in grams, is required for complete reaction of 25.0 grams of glucose? What masses of carbon dioxide and water, in grams, are formed? So, pause this video and see We're told that glucose, C6H12O6, reacts with oxygen to give carbon dioxide and water. We're not referring to the chemical formulas of the chemicals at the end, just to the chemical equation at the top and the coefficients. The way I think you're interpreting it is that moles = atoms in each compound, but that's not what we're dealing with here. Same idea for the water, the same 1:1 ratio. And to find moles of carbon dioxide from the moles of oxygen he uses a 6:6 ratio (really just a 1:1 ratio) from the chemical equation. So when Sal finds the moles of glucose from the grams of glucose, he multiples the moles of glucose by 6 to find the moles of oxygen since they are related to each other by a 1:6 ratio in the balanced chemical equation. One mole of glucose reacts with 6 moles of oxygen to produce 6 moles of carbon dioxide and 6 moles of water. When he says the moles of oxygen gas are equal to carbon dioxide and water, he means that in the chemical equation they all have a coefficient of 6. 11:11, he is using the coefficients of the balanced chemical equation at the top.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
